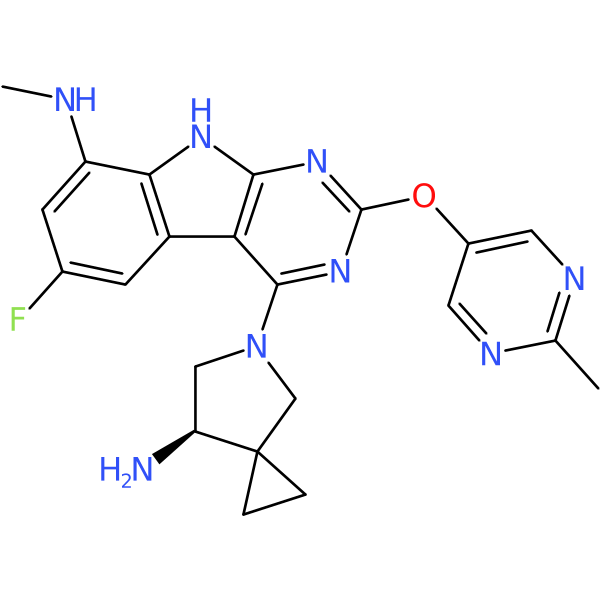
Compound ID | 137
GP-4
| Agent Type: | Synthetic; Small molecule; Direct acting; |
| Mechanism of action: | DNA synthesis inhibitor. Bacterial topoisomerase inhibitor |
| Institute where first reported: | Cubist Pharmaceuticals (Merck) USA |
| Year first mentioned: | 2014 |
| Development status: | Experimental |
| External links: | |
| Structure link: | https://pubchem.ncbi.nlm.nih.gov/compound/66560933 |
| Citations: |
|