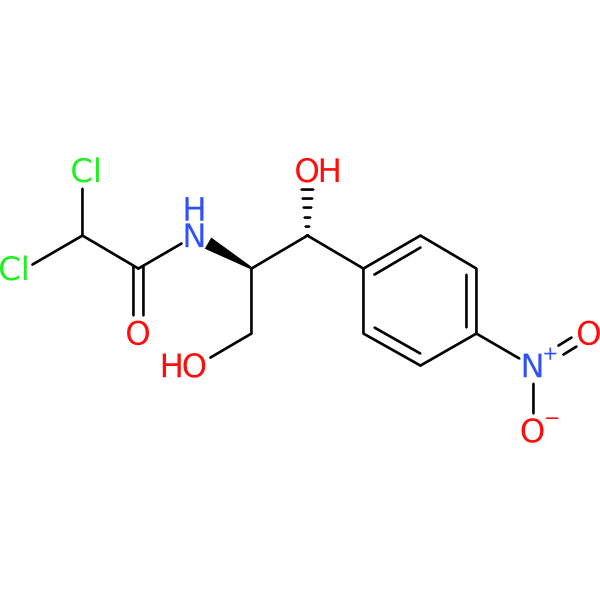
Compound ID | 1989
Chloramphenicol
Synonym(s): Chloromycetin
Class: Phenicol
| Agent Type: | Natural product; Small molecule; Direct acting; |
| Spectrum of activity: | Gram-positive & Gram-negative |
| Mechanism of action: | Protein synthesis inhibitor. Binds to 50S ribosomal subunit inhibiting the attachment of transfer RNA to the A site |
| Target Pathogen: | Broad spectrum activity |
| Description: | Natural product from Streptomyces venezuelae; developed for systemic and topical applications; has known adverse effects/toxicity (ex: bone marrow toxicity) |
| Institute where first reported: | Parke, Davis & Company; Yale University |
| Year first mentioned: | 1947 |
| Highest development stage: | Approved by FDA in 1949 but with restricted/regulated use |
| Development status: | Approved |
| External links: | |
| PubChem link: | https://pubchem.ncbi.nlm.nih.gov/compound/5959 |
| Guide to Pharmacology: | chloramphenicol |
| Citations: |
|