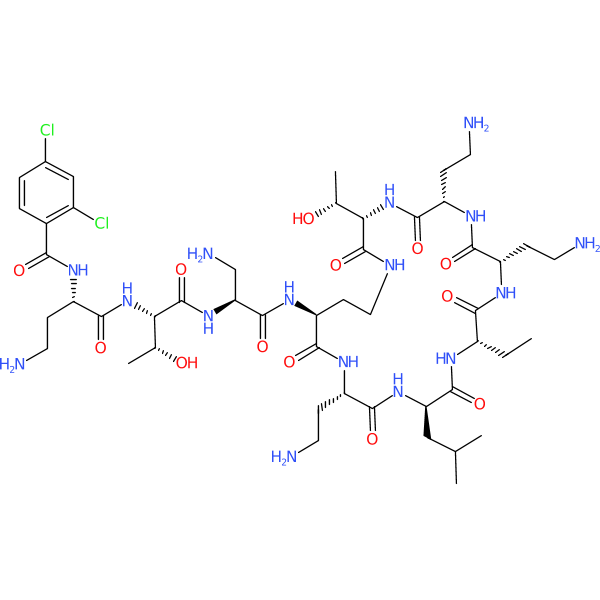
Compound ID | 2239
Soralimixin
Synonym(s): QPX9003 | F365 | F 365 | F-365
Class: Lipopeptide
| Agent Type: | Semisynthetic; |
| Spectrum of activity: | Gram-negative |
| Mechanism of action: | Unknown |
| Target Pathogen: | Active against Pseudomonas aeruginosa and Acinetobacter baumannii |
| Description: | Synthetic compound; rationally-designed derivative of polymyxin; reduced nephrotoxicity |
| Institute where first reported: | Qpex Biopharma, Inc.; Brii Biosciences; Monash University |
| Year first mentioned: | 2022 |
| Highest development stage: | Phase 1 (NCT04808414) |
| Development status: | Active |
| External links: | |
| Structure link: | https://pubchem.ncbi.nlm.nih.gov/compound/176507621 |
| Guide to Pharmacology: | soralimixin |
| Citation: | https://www.nature.com/articles/s41467-022-29234-3 |