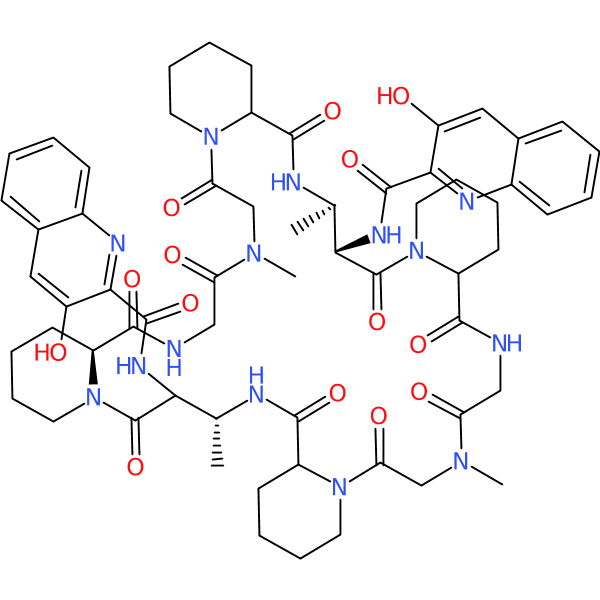
Compound ID | 2706
Quinaldopeptin
Class: Natural product antibiotic
| Agent Type: | Natural product; Small molecule; Direct acting; |
| Spectrum of activity: | Gram-positive & Gram-negative |
| Mechanism of action: | DNA synthesis inhibitor. Binds and intercalates into DNA |
| Target Pathogen: | Active against Staphylococcus aureus, Staphylococcus epidermidis, Streptococcus pyogenes, Streptococcus faecalis, Clostridium difficile, Clostridium perfringes, Propionibacterium acnes, Escherichia coli, and Klebsiella pneumoniae |
| Description: | Natural product from Streptoverticillium album strain Q132-6; a cyclic chromopeptide/ quinomycin-type antibiotic; shows antitumor activity towards P338 leukemia |
| Institute where first reported: | Bristol-Myers Research Institute, Ltd., Tokyo Research Center, Japan |
| Year first mentioned: | 1990 |
| Development status: | Experimental |