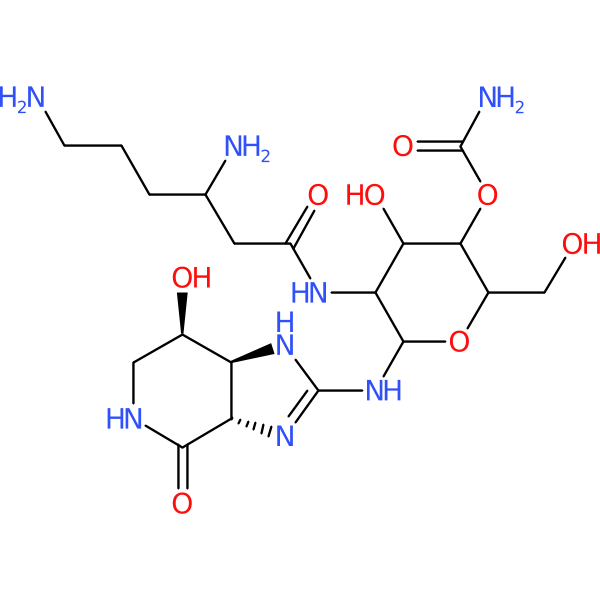
Compound ID | 2747
Streptothricin
Class: Streptothricin
| Agent Type: | Natural product; Small molecule; Direct acting; |
| Spectrum of activity: | Gram-positive & Gram-negative |
| Mechanism of action: | Protein synthesis inhibitor |
| Target Pathogen: | Broad-spectrum activity including Mycobacterium tuberculosis |
| Description: | Natural product from soil Actinomyces; shows delayed nephrotoxicity (~48 h after administration in mouse); small clinical trial in humans performed by Merck showed nephrotoxicity is reversible |
| Year first mentioned: | 1942 |
| Development status: | Inactive |
| Reason dropped: | Showed nephrotoxicity in human trials |