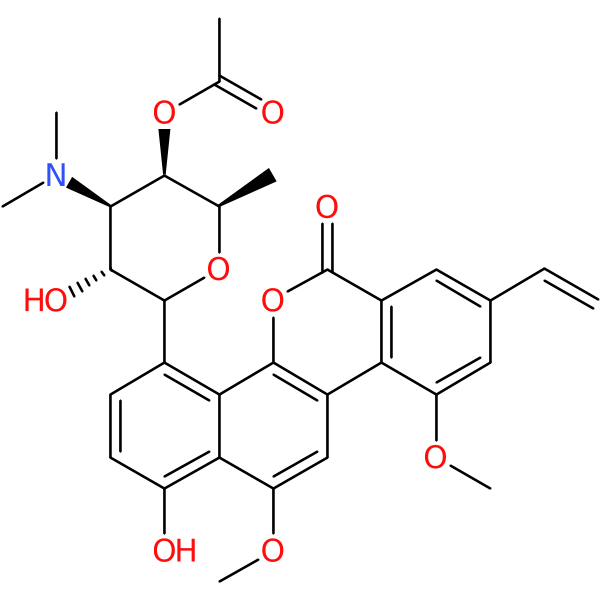
Compound ID | 3218
Ravidomycin
Synonym(s): AY-25,545 | AY 25,545 | AY-25545+N3226 | Deacetylravidomycin N-oxide
Class: Natural product antibiotic
| Agent Type: | Natural product; Small molecule; Direct acting; |
| Spectrum of activity: | Gram-positive & Antimycobacterial |
| Mechanism of action: | DNA synthesis inhibitor |
| Target Pathogen: | Active against Staphylococcus aureus (including penicillin-resistant strain), Streptococcus faecalis, and Mycobacterium tuberculosis var hominis |
| Description: | Natural product from Streptomyces ravidus; shows antitumor effect; aminoglycoside scaffold |
| Institute where first reported: | Department of Microbiology, Ayerst Research Laboratories |
| Year first mentioned: | 1983 |
| Development status: | Experimental |
| External links: | |
| PubChem link: | https://pubchem.ncbi.nlm.nih.gov/compound/126647 |
| Citations: |
|