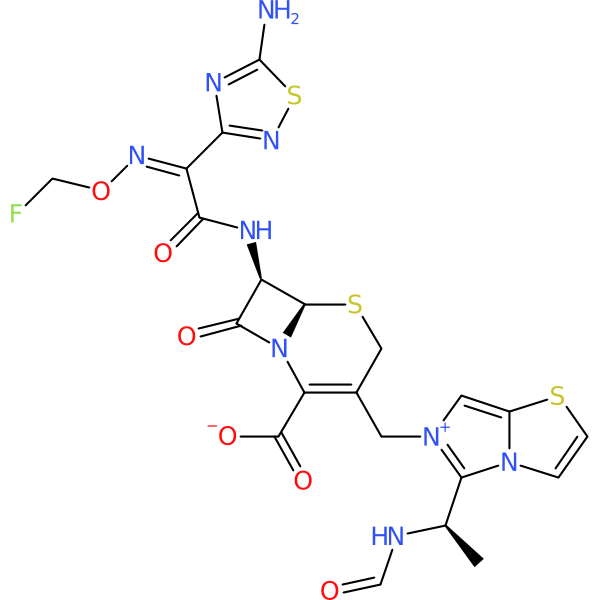
Compound ID | 432
CP6679
Synonym(s): CP-6679 | CP 6679 | ME1209
Class: Beta-lactam
| Agent Type: | Semisynthetic; Small molecule; Direct acting; |
| Spectrum of activity: | Gram-positive & Gram-negative |
| Mechanism of action: | Cell wall synthesis inhibitor |
| Propensity to select resistant mutants: | Lower propensity to select for resistant strains than ceftazidime |
| Description: | Injectable cephalosporin; Yoshida T, Ida I, Kurazono M, et al. ME1209 (CP6679), a new parenteral Cephalosporin II. In vitro and in vivo activity against Pseudomonas aeruginosa. 38th Intersci Conf Antimicrob Agents Chemother. 1998. |
| Institute where first reported: | Meiji Seika Kaisha, Japan |
| Year first mentioned: | 1995 |
| Development status: | Experimental |
| External links: | |
| PubChem link: | https://pubchem.ncbi.nlm.nih.gov/substance/273351054 |
| Citation: | https://doi.org/10.1007/s101560200024 |