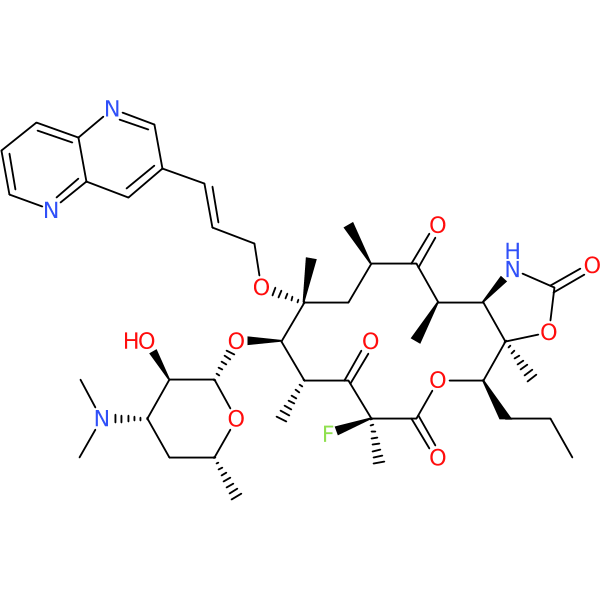
Compound ID | 598
JNJ-17155528
Synonym(s): JNJ 2
Class: Macrolide
| Agent Type: | Semisynthetic; |
| Spectrum of activity: | Gram-positive |
| Mechanism of action: | Unknown |
| Description: | Synthetic compound, derived from 15-methylerythromycin A |
| Institute where first reported: | Johnson & Johnson Pharmaceutical |
| Year first mentioned: | 2002 |
| Development status: | Experimental |
| Reason dropped: | Likely to be due to the failure of Ketek (Telithromycin) |
| External links: | |
| Citations: |
|