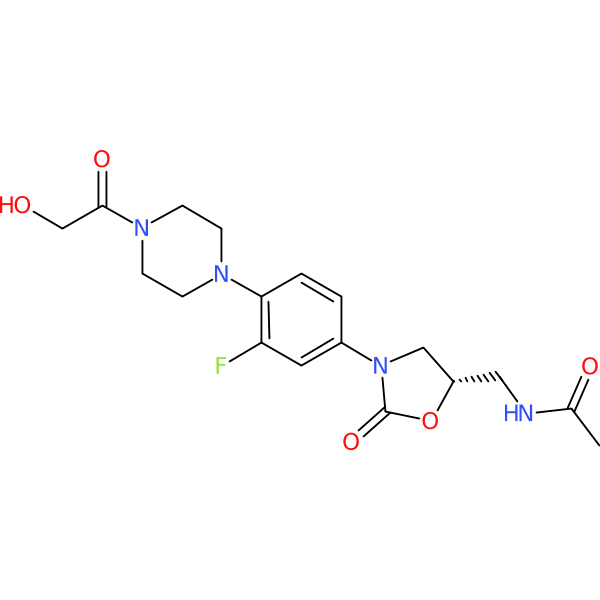
Compound ID | 757
Eperezolid
Synonym(s): PNU 100592 | U 100592 | U100592 | (S)-N-[[3-[3-fluoro-4-[4-(hydroxyacetyl)-1-piperazinyl]- phenyl]-2-oxo-5-oxazolidinyl]methyl]-acetamide
Class: Oxazolidinone
| Agent Type: | Synthetic; Small molecule; Direct acting; |
| Spectrum of activity: | Gram-positive |
| Mechanism of action: | Protein synthesis inhibitor. Binds to the 50S ribosomal subunit |
| Description: | A fluorinated oxazolidinone; Pawsey SD, Harry JD, Stalker DJ. 1st administration of a new oxazolidinone antibiotic (U-100592) to man. 35th-ICAAC 1995;15 |
| Institute where first reported: | Pharmacia & Upjohn (Pfizer, USA) |
| Year first mentioned: | 1995 |
| Highest development stage: | Phase 3 |
| Development status: | Inactive |
| Reason dropped: | Less effective than linezolid; development of resistance observed |
| External links: | |
| PubChem link: | https://pubchem.ncbi.nlm.nih.gov/compound/73214 |
| Guide to Pharmacology: | eperezolid |
| Citations: |
|